
In late January, the Parkinson Society Southwestern Ontario offered a webinar on Advanced Therapies for Parkinson’s Disease (PD), featuring speaker Devin Hall, MD. She gave an overview of treatments that sometimes used for later-stage PD, such as Deep Brain Stimulation, apomorphine injections, and levodopa intestinal gel (Duopa), discussing their benefits and drawbacks. We at Stanford Parkinson’s Community Outreach listened to the webinar and are sharing our notes.
To watch this webinar and others from the Parkinson Society Southwestern Ontario, follow this YouTube link.
If you have questions about this webinar, you can reach out to Kaitlin Garbutt, Community Development Coordinator with the Parkinson Society Southwestern Ontario, at kaitlin.garbutt@parkinsonsociety.ca.
To learn more about Deep Brain Stimulation (DBS), see the Stanford Parkinson’s Community Outreach webpage on this topic.
For resources on managing late-stage PD, visit the Stanford Parkinson’s Community Outreach website.
Now… on to our notes from the webinar.
– Lauren
. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
Advanced Therapies for Parkinson’s Disease
The Parkinson Society Southwestern Ontario
Webinar on January 21, 2020
Notes by Lauren Stroshane, Stanford Parkinson’s Community Outreach
Dr. Devin Hall is a Movement Disorder Neurologist at Hamilton General Hospital and Assistant Professor of Neurology at McMaster University in Ontario, Canada. She provided an overview of the characteristics of advanced PD, then discussed possible treatment options.
What is advanced PD?
As most people know, the typical signs of early PD include tremor, slowness, stiffness, and sometimes imbalance – known as “motor symptoms.” As the disease progresses, motor symptoms progress, and may include falls, freezing of gait, and motor fluctuations, which are discussed below. “Non-motor” symptoms may also become more prominent, but were not the focus of Dr. Hall’s presentation. She emphasized that motor fluctuations are the target for the advanced therapies she was discussing. They are often disabling, unpredictable, and difficult to treat.
Motor fluctuations can include a range of issues:
- Dyskinesias – uncontrolled writhing movements triggered during the medication cycle
- Partial or delayed on – medications do not fully “kick in” or take longer to kick in
- Early wearing off – medications do not last as long as they used to between doses
- Sudden off – medications suddenly seem to stop working
- Dose failure – feeling as though one skipped a dose of medication, despite taking it
Here is an excellent fact sheet from the American Parkinson Disease Association (APDA) on motor fluctuations, in PDF format.
Managing the narrowing therapeutic window
As PD progresses, the “therapeutic window” – in which the brain is getting enough dopamine to move well, but not too much dopamine that can trigger dyskinesias or other side effects – narrows substantially, making control of motor symptoms increasingly difficult.
The standard treatment for wearing off typically involves trying a number of strategies to see which works best for the individual: shortening the time between doses of levodopa, for instance, taking levodopa four times a day instead of three; adding or switching to controlled-release levodopa; or adding another medication from a different drug class (such as pramipexole, entacapone, or rasagiline). Sometimes these measures can be quite effective.
For those who experience “peak dose” dyskinesias – uncontrolled movements that occur when the levodopa dose is at its peak effect – adding the medication Amantadine can sometimes be helpful.
Eventually, these measures may be insufficient to combat motor fluctuations, which can significantly worsen quality of life. Going out in public, enjoying nature, participating in hobbies, exercise, or social events can all become impaired by the unpredictability of motor fluctuations. When this is the case, it may be time to consider advanced therapies. Dr. Hall discussed apomorphine, Deep Brain Stimulation, and levodopa gel given through an intestinal pump (Duopa).
What is apomorphine?
The class of drugs called dopamine agonists includes some medications that are used for Parkinson’s disease, including pramipexole (Mirapex), rotigotine (Neupro), and ropinirole (Requip). Apomorphine (brand name Apokyn) also falls within this category, but has a more specialized use: treating sudden “off” episodes. Approved by the FDA in 2015, it is given as a subcutaneous injection just under the skin, similar to insulin, and acts very quickly. For those who do not wish to pursue more invasive measures such as Deep Brain Stimulation or an intestinal pump, or are not candidates for other reasons, apomorphine injections can be a useful tool for managing motor fluctuations. You can read more about apomorphine here.
Typically, your neurologist needs to have already optimized your existing PD medications, meaning they have already tried various options within the standard treatments, before you try apomorphine. To get started, typically your neurologist will start you on an anti-nausea medication one to three days before initiating apomorphine injections. Usually you will be asked to hold your PD medications for 12 to 24 hours ahead of time, so that you will arrive “off” medications on the day of. Once you arrive to the clinic, your blood pressure, heart rhythm, bloodwork, and symptoms will be checked. Then the staff will proceed to give you an injection of apomorphine every one to two hours until the correct dose is found. Before going home, you should be instructed in how to perform the injections yourself.
Possible side effects include nausea and vomiting, which are common, and can usually be managed with ant-nausea medication; skin reactions at the injection site, such as minor bruising; dizziness, typically caused by low blood pressure, which can be a side effect of apomorphine; leg swelling; episodes of drowsiness or falling asleep; impulse control disorder, which can lead to compulsive behaviors such as gambling or shopping; and hallucinations.
What is carbidopa-levodopa intestinal gel (Duopa)?
Approved in 2015 by the FDA, Duopa is a new formulation of the standard PD medication levodopa. Instead of a pill being swallowed, levodopa is suspended in a gel and then pumped directly into the intestines. This gel allows the levodopa to be absorbed more completely and consistently by the body, while the pump can keep a steady rate of medication. Together, these factors promote longer, steadier “on” time, since there is less cycling between doses of oral medication, and the ability to fine-tune dosing more precisely. Duopa is best for those who respond well to oral levodopa but spend at least a quarter of their waking hours feeling “off”.
The pump necessitates surgical creation of a small hole – called a stoma – through the abdominal wall into the intestine. Those who have pre-existing intestinal, stomach, or pancreatic issues may not be able to receive Duopa. Untreated liver, heart, lung, kidney, blood, or hormone disease are also contraindications for this procedure. The tube insertion surgery is performed by a gastroenterologist, who must coordinate care with your neurologist.
Possible side effects include those that apply to oral levodopa, such as nausea, dizziness, dyskinesias, constipation, and hallucinations. Other side effects pertain to the intestinal tube procedure and can include abdominal pain; redness, swelling, or infection at the tube insertion site; and leakage from the stoma.
You can read more about Duopa here.
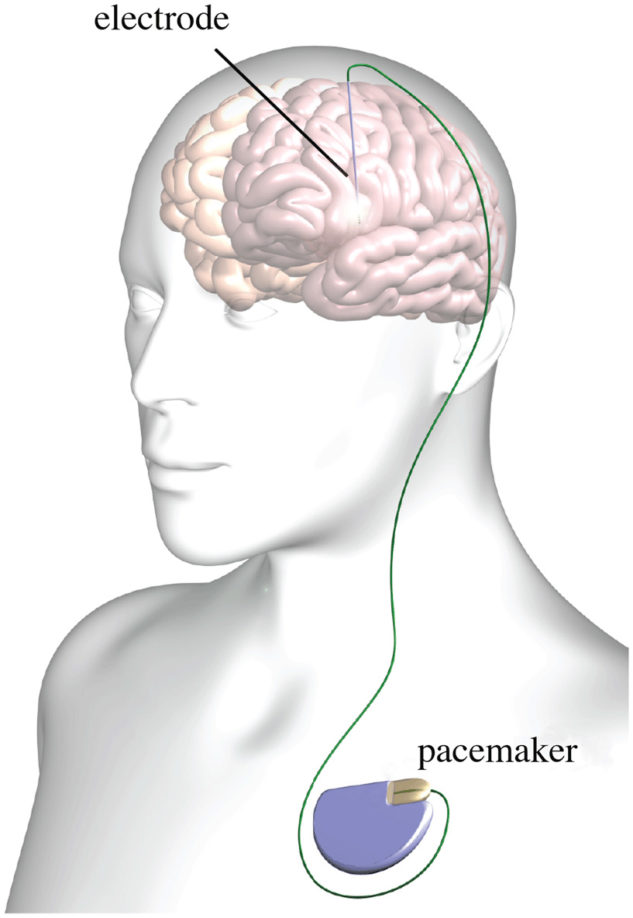
What is Deep Brain Stimulation (DBS)?
Deep Brain Stimulation involves the surgical implantation of tiny electrodes into the areas of the brain affected by PD (hence “deep brain”), and a small battery placed in the chest. This can be in either one or both sides of the brain, depending on symptoms. Small doses of electrical current stimulate the brain and help to treat motor symptoms, medication-induced dyskinesias or other side effects, tremor that does not respond to medication, and motor fluctuations.
The Stanford Parkinson’s website has many excellent resources on DBS, to learn more.
DBS is not only used for those with advanced PD, but is now often implanted earlier in the disease as well; it has been around for twenty years as a therapy for PD. While the battery will need to be replaced over the years (how often depends on the specific model), the electrodes themselves continue to work indefinitely, even in late stages of the disease. DBS can be very helpful for treating the motor symptoms of PD, but does not treat non-motor symptoms, balance, or freezing of gait.
To be a candidate for the surgery, you should have had PD for at least 3 to 4 years, and may be experiencing motor fluctuations despite optimization of your oral medications. Ideally your symptoms should respond to levodopa. Those with significant cognitive impairment; uncontrolled comorbidities such as heart, lung, liver, kidney, blood, or hormonal diseases; or significant psychiatric disorder may not qualify for the surgery. Surgical centers typically require a substantial evaluation head of time to ensure you are a good candidate. This often involves testing your motor symptoms both on and off medication, cognitive testing, and a brain MRI.
The surgery to implant the brain electrodes itself place in an operating room, either awake or asleep. Then the battery/stimulator is implanted in the chest on a different day, usually asleep. After some recovery time has passed, there is a clinic visit to turn the device on and subsequent programming sessions.
Possible adverse effects of DBS include serious complications of brain surgery, such as infection or stroke, as well as transient side effects during programming sessions. One advantage of the device is that it can be reprogrammed over time, or turned off, making it a reversible procedure.
Possible future therapies
Dr. Hall mentioned some new and exciting therapies being explored for symptom management of PD.
Focused ultrasound has been approved by the FDA for treatment of essential tremor, and is being studied for PD as well. This non-invasive procedure uses ultrasound technology to create a small, targeted lesion in the brain that can treat motor symptoms of PD. It is far less invasive than Duopa or DBS, but it is not reversible, and is unilateral only – meaning only one side of the brain can be targeted.
Spinal cord stimulation is being studied as a possible treatment for freezing of gait (FOG), an often-disabling gait disturbance that can occur in PD. A stimulator device is surgically implanted in the spine and later programmed to find the optimal settings for each patient, to help improve their gait. This therapy is already available as a treatment for pain.
Question & Answer Session
Q: How long does DBS work? What is the life expectancy of the device itself?
A:It does not stop working; non-motor and motor symptoms will continue to progress over time, but programming can be adjusted and the device will continue to provide some benefit for motor issues. Depending on the type of device, battery will need to be replaced in 3-5 years for a non-rechargeable device, or up to 8 or 9 years for rechargeable DBS systems.
Q: Can DBS cause mania and how to manage this if it occurs?
A: This is not a typical side effect of DBS but depending on the parameters, the stimulation can cause mood changes. This is assessed during a programming session, and the program can usually be changed to avoid these side effects. It may be worth evaluating your oral medications as well, to see if these could be causing mania as a side effect.
Q: I’ve been approved for DBS surgery, but should I wait for focused ultrasound (FUS) to be approved for PD and get that instead?
A: It is unclear how much longer it will take for FUS to be approved for PD. There are advantages and disadvantages to both procedures. Keep in mind that FUS is only available for one side of the brain (and body), and that the procedure is permanent – not reversible or reprogrammable, like DBS. On the other hand, FUS is also much less invasive than DBS surgery. It is a highly individual decision and should be discussed with your neurologist.
Q: Can beta blockers increase the risk of developing PD?
A: Nothing has been definitively proven about a connection between beta blocker use and risk of developing PD. Keep in mind that having PD does not prevent you from developing other conditions, such as high cholesterol or high blood pressure, which can contribute to stroke. It is best to talk with your physician(s) before making any changes to your medications.
Q: Does DBS increase the likelihood of PD patients getting dementia?
A: DBS does have risks to worsen memory and thinking; for someone who already has some impairment, theoretically this could push them over into real cognitive impairment and affect their functional ability. This is the reason for cognitive testing ahead of time, to make sure the patient, family, and care team are educated about all the possible risks of the procedure.