
On January 10, 2026, Stanford Medicine hosted a seminar on “Gut-Brain & Parkinson’s Disease”, exploring how the gastrointestinal tract may hold the key to both the origin and early detection of the disease. This seminar shared the latest between bioengineering and clinical neurology, presenting a compelling case for the “Body-First” hypothesis—the idea that Parkinson’s pathology may begin in the gut due to environmental triggers before reaching the brain. The featured Stanford speakers in different fields included movement disorder neurologists Dr. Kathleen Poston and Dr. Bianca Palushaj, neurogastroenterologist Dr. Leila Neshatian, and researcher Yasmine Kehnemouyi, PhD candidate, and ended with a panel moderated by bioengineer Dr. Todd Coleman. These are notes from the seminar.
Together, the speakers shifted the focus from a brain-only model to a view of the “little brain” in the gut, emphasizing that non-motor symptoms like constipation are not just side effects, but central signals of the disease process. There was a great deal of detailed and in-depth information covered by each of the speakers, reflecting the complex relationship between our digestive health and neurological function.
Here are quick summaries of the four speakers’ presentations (with links to their slides) and the panel:
- “Understanding the connection between the gut and brain in Parkinson’s Disease” (Dr. Kathleen Poston): There is some evidence that suggests Parkinson’s begins in the gut where toxins influence changes that eventually leads to protein aggregation that could travel to the brain via the vagus nerve. Because constipation often precedes motor symptoms by 20 years, researchers are looking at routine colon biopsies (done during standard colonoscopies at age 45) as a way to find PD markers early.
- “Measuring Cholinergic Dysfunction in Parkinson’s Disease” (Yasmine Kehnemouyi, PhDc): While many researchers focus on dopamine, the gut-brain axis relies on the cholinergic (acetylcholine) system, which degenerates early in the disease process. New non-invasive technology like EEG, EKG, and stomach sensors (EGG) to monitor the body’s “pre-digestion” response to food to identify this nerve dysfunction before a clinical diagnosis is possible.
- “Rethinking the Gut in Parkinson’s Disease: A declining microbial ecosystem?” (Dr. Bianca Palushaj): Using a new “Absolute Abundance” counting method, Stanford found that PD patients aren’t just overgrown with “bad” bacteria—they have less bacteria in their gut overall, and are severely depleted of “good” bacteria, several of which produce protective short-chain fatty acids. The PD gut appears to be a much less welcoming place for healthy microbes to live, meaning that restoring balance may require not just adding “good” bacteria, but rebuilding the environment that allows them to survive and thrive.
- “Moving Things Along: Constipation in Parkinson’s- Tips, Tools, and Research Updates” (Dr. Leila Neshatian): Most Parkinson’s-related constipation is actually a muscle coordination failure (evacuation dysfunction) that requires specialized biofeedback physical therapy rather than standard laxatives. Additionally, bacterial overgrowth in the small intestine (SIBO) can “steal” levodopa, preventing the medication from reaching the brain effectively.
- Panel: To maximize absorption, levodopa should be taken one to two hours away from high-protein meals, though maintaining protein intake (1g protein per kg of body weight) and daily exercise remains essential for building brain resilience and muscle mass. Individualizing the timing of medication and carbohydrates can help manage nausea without compromising the drug’s efficacy.
For those who live near Stanford and want to participate in gut-brain and Parkinson’s research, please complete this online enrollment form:
If you have questions, email the Stanford Gut-Brain Research Study team at GutBrainResearch@stanford.edu
This seminar was not recorded as it included information about unpublished research. But, I attended and took detailed notes. See below.
– Elizabeth
“Gut-Brain and Parkinson’s Disease” Seminar
Seminar Host: Stanford School of Medicine Poston Lab
Seminar Date: January 10, 2026
Speakers were:
- Dr. Kathleen Poston: Professor of Neurology and Movement Disorders at Stanford. She serves as a lead investigator for the NIH Gut-Brain PD Consortium.
- Yasmine Kehnemouyi, PhD Candidate: Graduate researcher with Dr. Todd Coleman Lab at Stanford, specializing in multi-organ electrical monitoring and cholinergic dysfunction.
- Dr. Bianca Palushaj: Movement Disorders Neurologist at Stanford with research focusing on absolute microbial abundance and the gut ecosystem.
- Dr. Leila Neshatian: Director of Neurogastroenterology and Motility at Stanford, specializing in clinical management of GI dysfunction and pelvic floor disorders.
- Dr. Todd Coleman (Panel Moderator): Professor of Bioengineering at Stanford, whose lab develops the non-invasive sensing technologies used in this research.
Summary by: Elizabeth Wong, Stanford Parkinson’s Community Outreach
“Understanding the connection between the gut and brain in Parkinson’s Disease”
Dr. Kathleen Poston

PD symptoms span from non-motor (preceding diagnosis) to motor (classical) to other non-motor.
- Early Non-Motor: Sleep disorders, depression, anxiety, and constipation.
- Classical Motor: Slowness (bradykinesia), stiffness (rigidity), and tremor.
- Other Non-Motor: Apathy (lack of interest) and blood pressure fluctuations.
Symptoms can overlap, appear in different orders, or come and go. No two patients are identical; some never develop tremors, while others never experience GI issues. Neither patients nor doctors can currently predict the severity or timing of specific symptoms.
Pathology: Broadly, Parkinson’s is considered a “Lewy body disease.” In the brain, we observe Lewy bodies—aggregates of the protein alpha-synuclein—which can be identified through specialized staining of nerve cells. Beyond motor nerves, these Lewy bodies are found in sensory, cognitive, neuropsychiatric, and gut-related nerves.
Historically, constipation has been the only gastrointestinal (GI) symptom recognized on clinical charts. However, GI dysfunction in Parkinson’s is far more detailed and expansive than constipation alone. Because these GI issues often appear years before motor tremors, they provide us with critical insight into the role of the GI tract and offer a potential pathway to earlier diagnosis.
PD affects the entire GI tract:
- Mouth/Throat: Excessive drooling (sialorrhea), dental carries, and swallowing problems (dysphasia).
- Esophagus: Motility issues occurring both in the back of the throat and further down the tube.
- Stomach: Significant changes in stomach acid levels and food movement.
- Small & Large Intestine: Challenges with transit speed and nutrient absorption, which can be quantified through new technological methods.
Traditional vs. Modern Diagnostic Approaches in Neurology:
- Traditional Screening: Primarily relies on a massive array of subjective questionnaires. The GI world has many specialized questionnaires for these specific symptoms.
- Management History: Historically, PD GI symptoms have been managed by “borrowing” general therapies from the non-PD GI world.
- Research is now focused on quantitative techniques (innovative wearables and biomarkers) to develop therapies specific to the unique cellular environment of a Parkinson’s gut.
The “Body-First” Hypothesis & Environmental Interaction
- In the last decade, our understanding has shifted from viewing Parkinson’s as a “brain-first” disorder to considering a “body-first” model. This hypothesis suggests that Parkinson’s may be triggered by environmental exposures. This is an “active idea” with data to both support and refute it; it is not yet a proven fact.
- Mechanism: Environmental factors (air pollution, pesticides, toxins) interact with the GI lining. This may cause inflammation and cellular changes that eventually spread to the brain.
- Pathophysiology:
- Thinning Mucosa: A breakdown in the protective layer of the GI tract.
- Leaky Gut: The breakdown of “tight junctions” (connections between cells/nerves), allowing toxins into the bloodstream that should be filtered out.
- Inflammatory Cascade: This breakdown may act as the “light at the end of the fuse,” starting an inflammatory reaction that travels to the brain.
The PD Gut-Brain Consortium & Research Methodology:
- The Consortium: An NIH-funded team of six sites across the U.S. using a unified approach on how to study patients.
- Household Study Logic: By studying PD patients alongside their household members (spouses/partners), researchers can control for environmental variables like shared diet and water sources.
- Next-Generation Analysis: Older methods identified only 4–5 cell types in a biopsy. New machine-learning-driven techniques can identify dozens of distinct cell types by color-coding every cell in a high-resolution image.
- Comparison Data: Research shows that the proportionality of cell types in a PD colon is distinct from both “neurologically healthy” individuals and those with Inflammatory Bowel Disease (IBD).
- The Vision: Because colonoscopies are now standard at age 45, these routine screenings can be used to identify PD-specific cellular changes years before the first motor tremor starts. This would open the door to preventative therapies.
“Measuring Cholinergic Dysfunction in Parkinson’s Disease”
Yasmine Kehnemouyi, PhD

Limitations of Current PD Characterization
- Standard Definition: PD is primarily defined as the second most common neurodegenerative disorder and the most common movement disorder, caused by the death of dopamine-producing neurons in the substantia nigra.
- The Clinical Gap: Current therapies focus almost exclusively on this dopamine deficit. However, PD is a complex, multi-system pathology involving non-motor systems that produce:
- Cognitive impairment
- GI dysfunction
- Anosmia/Hyposmia (loss of smell)
- Sleep disorders
- Proposal: Parkinson’s should not be categorized merely as a movement disorder, but as a holistic systemic failure of multiple neurotransmitter pathways.
Cholinergic Degeneration (The Acetylcholine Pathway)
- Gut-Brain Axis Anatomy:
- Enteric Nervous System (ENS): containing the stomach.
- Central Nervous System (CNS): containing the brain.
- Autonomic Nervous System (ANS): controls a lot of the involuntary physiological processes such as pupil dilation and constriction, breathing, health rate gastric motility and emptying.
- Vagus Nerve: The largest nerve in the autonomic nervous system; the bidirectional “highway” between the ENS and CNS.
- The Role of Acetylcholine: While dopamine dominates the brain, acetylcholine is the primary chemical messenger for the gut-brain axis.
- Pattern of Spread: New research indicates degeneration starts in the acetylcholine-producing neurons of the stomach. This cholinergic degeneration spreads through the vagus nerve, affecting areas throughout the autonomic nervous system, eventually reaching the central nervous system (brainstem).
- The DMV (Dorsal Motor Nucleus of the Vagus): A critical nucleus in the brainstem that produces high volumes of acetylcholine. This area is highly susceptible to early degeneration, serving as a key “checkpoint” for disease progression.
Physiological Impact of Cholinergic Degeneration in the ANS and ENS
> Autonomic Nervous System (ANS) Failure
- The ANS controls involuntary processes (breathing, heart rate, pupil dilation).
- Gastroparesis: Loss of acetylcholine in the vagus nerve leads to the “slow-moving” of food and drink through the stomach, causing bloating and nausea.
> Enteric Nervous System (ENS) Failure
- The ENS coordinates the complex muscle movements required for bowel movements.
- Constipation: A direct result of reduced acetylcholine in the enteric system, leading to worsened GI motility.
The Prodromal (Pre-Motor) Window
- Symptoms such as REM Sleep Behavior Disorder (RBD), hyposmia (smell loss), and constipation appear decades before motor symptoms.
- The Clinical Issue: By the time motor symptoms allow for a traditional diagnosis, the efficacy of disease-modifying therapeutics is significantly decreased. This led the researchers to look at how to measure cholinergic pathways in a non-invasive way, which led them to the DMV and developing a way to activate the DMV and measure its activity.
The Cephalic Phase
The Cephalic Phase is the pre-digestion phase, accounting for 20% of total digestive activity before food even enters the mouth. It is regulated entirely by cholinergic activity through the DMV.
The Mechanism of the Cephalic Phase:
1. Sensory Input: Sight, smell, or even the sound of food sizzling activates the cerebral cortex.
2. Brainstem Signaling: The cortex signals the DMV to prepare the body.
3. Organ Activation: The DMV triggers salivary glands, the heart (rate changes), and the stomach.
4. Preparatory Contractions: The stomach initiates neuromuscular contractions to prepare for incoming food.
The previous “Sham Feeding” method used to measure the cephalic phase had limitations. Prior research used “chew and spit” methods to measure the cephalic phase, where participants would chew something, then spit it out to engage this sense of taste and then a blood sample to measure pancreatic polypeptide that is secreted from the pancreas. These were invasive (requiring blood draws), relied on taste, and ignored the multi-organ nature of the response (brain, heart, and swallowing muscles).
The Stanford Engineering Solution: Multi-Organ Electrical Measurement. To measure cholinergic degeneration non-invasively, the Coleman and Poston Labs have developed a multimodal monitoring system to capture signals during the cephalic phase.
Measurement Modalities:
- EEG (Electroencephalography): Measures cortical brain activity in response to food stimuli.
- EMG (Electromyography): Measures electrical activity in salivary glands and swallowing muscles.
- EGG (Electrogastrogram): A specialized electrophysiological tool using sensors on the surface of the abdomen to measure: EKG (heart rate variability) and neuromuscular activity (the preparatory “muscle rhythm” of the stomach).
The “Hunger State” Protocol Study Design. The study uses a three-tier design to identify the “cholinergic signature” of PD:
Baseline Period: Establishing the subject’s resting electrical state.
Hunger State Engagement: Utilizing personalized food preferences (via prior surveys) to present the subject with sight and smell stimuli.
Consumption Phase: Measuring the transition from “pre-digestion” to active digestion.
Research Goals & Recruitment:
- Goal: Move away from invasive lumbar punctures toward non-invasive, biology-based electrical biomarkers.
- Recruitment: Currently enrolling both Parkinson’s patients and age-matched healthy controls (specifically household controls to account for shared environments).
- Location: Studies are conducted in-clinic at Stanford, in the lab building, and through home-based monitoring.
Editor’s Note: To participate in this research at Stanford, contact Yasmine at ykehn97@stanford.edu, by phone at 301-221-1948, or via gutbrainresearch@stanford.edu.
“Rethinking the Gut in Parkinson’s Disease: A declining microbial ecosystem?”
Dr. Bianca Palushaj

The Microbiome
The microbiome refers to the trillions of organisms that live in us and on us. Microorganisms play a crucial role in numerous physiological and pathological processes.
The 40% Human Rule: By cell count, humans are only 40% human. Microbial cells (trillions of organisms) outnumber human cells, making the microbiome a primary driver of health outcomes.
Immune Centrality: 70% of the human immune system resides in the gut. The microbiome “trains” this system and influences its daily function.
Microbes can influence genes that are turned on and off, they metabolize what we eat and create metabolites which can actually influence gene expression.
Neurochemical Production: The gut is a major chemical factory for the brain –
- 90% of the body’s serotonin is produced in the gut.
- 50% of the body’s dopamine is produced in the gut.
The gut as a multi-layered environment:
- The Microbiota: Microbes (bacteria, etc.) existing in the gut lumen.
- The Mucus Barrier: A protective green “buffer” layer.
- The Gut Wall: The blue layer containing immune and nerve cells that constantly sense microbial products and inflammatory molecules.
Pathophysiology: The Fiber-Mucus-Inflammation Cycle
- The Role of Fiber: Microbes metabolize dietary fiber. When fiber intake is insufficient, microbes begin to digest the carbohydrates in the gut’s mucus layer as an alternative fuel source.
- Thinning of the Barrier: This “predatory” microbial behavior thins the mucus layer over time.
- Immune Activation: As the barrier thins, environmental toxins, bacteria, and inflammatory byproducts come into direct contact with the gut wall (where the nerve cells live). This triggers the inflammatory immune response suspected of lighting the “fuse” for Parkinson’s pathology.
Two approaches to microbiome research – Relative vs. Absolute Abundance:
> The “Relative Abundance” Error
- Standard Practice: Most studies report bacteria as a percentage of the total community.
- The Mathematical Trap: Because percentages must always add up to 100%, if one bacterial species decreases, another automatically appears to increase as a percentage, even if its actual number remains the same. This leads to misleading conclusions.
> Absolute Abundance (The Stanford Approach)
- The Technique: Conducted in the Bhatt Lab (Dr. Ami Bhatt at Stanford), this method measures the actual quantity of microbes.
- Findings (The Volcano Plot):
- Comparing the relative abundance approach to absolute abundance approach shows that different species become statistically significant. This has allowed us to make new associations between specific species and PD. It demonstrates the misleading conclusions that can be drawn from relative abundance.
Key Findings
Key findings in the Parkinson’s gut based on a study of 193 participants (including 79 household pairs to control for shared water, diet, and pets):
> The Depletion Trend
- Findings: In the Absolute Abundance data, there are significantly more depleted microbes (green dots on the plot) than enriched ones (red dots).
- PD is a Disease of Loss: The PD microbiome is characterized more by a loss of beneficial bacteria than by the overgrowth of “bad” bugs.
> Short-Chain Fatty Acid (SCFA) Deficiency
- The most depleted microbes in PD are those responsible for producing short-chain fatty acids (SCFA).
- Why SCFAs matter: They dampen inflammation and maintain the integrity of the gut barrier. Their loss directly contributes to “leaky gut.”
> Decreased Total Microbial Load
- The Discovery: People with PD have a decreased total microbial load (fewer bacteria overall) compared to healthy controls.
- Machine Learning (ML) Model: Stanford developed an ML model that can accurately predict this total load from samples, eliminating the need for direct, expensive measurement in future studies.
Clinical Implications
Clinical Implications of this research redefines the ecosystem showing that the gut is not just a collection of bacteria; it is an environment shaped by factors such as pH, oxygen levels, diet, motility, and others. Findings suggest the PD gut has become an ecosystem that is less supportive of microbial growth.
The Therapeutic Strategy: Simply taking a probiotic (adding one or multiple bugs) may fail if the environment is hostile. True restoration requires creating an environment (through diet, pH management, and motility) that allows beneficial bacteria to thrive.
Nature’s Example: Baby elephants eat the feces of adults to “seed” their gut with the specific microbes necessary to transition from milk to a plant-based diet, illustrating the necessity of a colonized, functional ecosystem.
“Moving Things Along: Constipation in Parkinson’s- Tips, Tools, and Research Updates”
Dr. Leila Neshatian
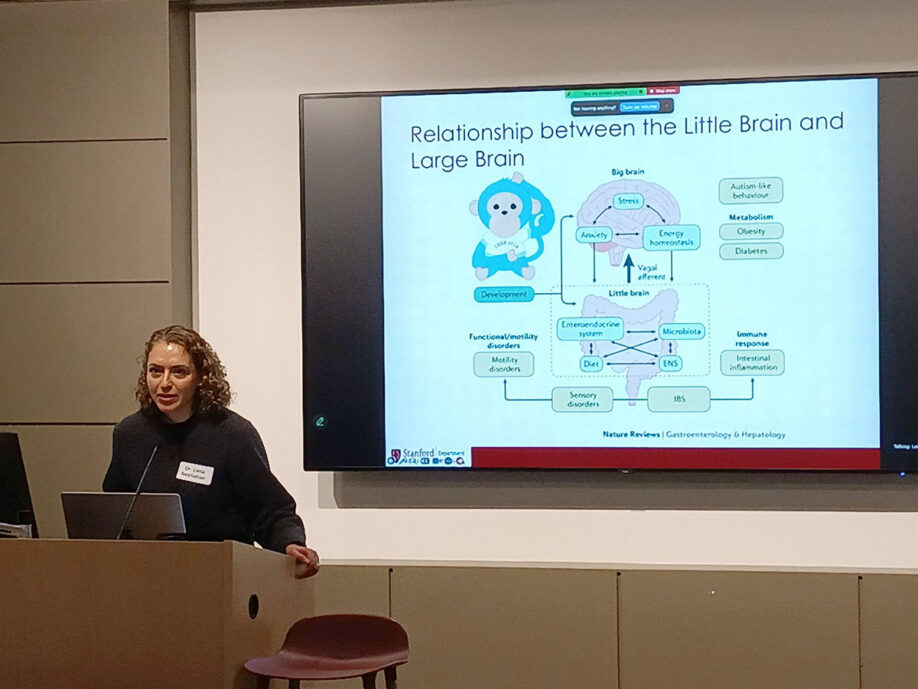
The “Little Brain” – The Enteric Nervous System (ENS):
- Independence of the CNS: ENS can be seen as the “Little Brain” because the nerves in the GI tract can sense and control their environment independent of the brain (Central Nervous System).
- Function: These nerves coordinate contractions and motility, a process heavily influenced by the bidirectional gut-brain axis and the microbiome.
- Clinical Impact: In PD, this “Little Brain” is compromised long before motor symptoms appear, leading to systemic GI failure.
Clinical Scope of Constipation in PD:
- Prevalence: More than 80% of PD patients report infrequent bowel movements, excessive straining, or the sensation of incomplete evacuation.
- Correlation with Severity: Patients with chronic constipation typically experience: worse motor symptoms (stiffness, slowness); increased non-motor symptoms (bladder dysfunction, blood pressure instability); worse overall disease prognosis.
- Etiology: Constipation in PD is multifactorial, stemming from dietary/hydration deficits; medication side effects (PD drugs often target acetylcholine or dopamine pathways, which inadvertently slow the gut); physiological failure (inefficient colonic contractions or muscle dysregulation in the pelvic floor).
Two Subtypes of Constipation
“Constipation” is an umbrella term for two distinct physiological problems (or subtypes) that require different treatments:
Problem #1 – Slow Transit Constipation
- The Issue: The colon itself is “lazy.” Contractions are weak or infrequent, leading to slow movement of stool.
- Diagnosis: Measured via Colonic Motility Tests.
- Standard Treatment: Medical management.
- First-line: Fiber and hydration.
- Second-line: Osmotic laxatives (Miralax, Magnesium salts) or stimulant laxatives.
- Advanced: Newer prescription secretagogues, enemas, or trans-anal irrigation.
Problem #2 – Evacuation (Defecatory) Dysfunction
- The Issue: A coordination failure of the anal sphincter and pelvic floor muscles. These muscles may be spastic (contracting when they should relax) or dysregulated.
- Prevalence in PD: Nearly 80% of PD patients with constipation have some form of evacuation disorder. Stanford’s internal data shows that 60% of severe cases that failed medical treatment were actually suffering from this subtype.
- Standard Treatment: Biofeedback Therapy (specialized physical therapy). Laxatives are largely ineffective for this condition because the problem is mechanical/coordination-based, not transit-based.
The Transit Phase – Advanced Diagnostic Tools & Emerging Tech for Motility Testing
- Sitz Marker Test (current standard): A capsule containing 24 radio-opaque markers. X-rays are taken over several days to track the markers’ progress. This is the most accessible and cost-effective test.
- MotilityCap™ (emerging): An ingestible capsule that senses pH (acidity) and temperature to track motility from ingestion to exit.
- Atmo® (emerging): A “gas-sensing” capsule that determines transit time based on the specific gas profiles found in different segments of the GI tract.
The Evacuation Phase – Advanced Diagnostic Tools & Emerging Tech for Functional Testing
- High-Resolution Anorectal Manometry: A flexible 5-inch catheter with sensors and a balloon is inserted into the rectum. It measures:
- Strength: Ability to hold stool and avoid accidents.
- Coordination: Ability to relax the sphincter while increasing rectal pressure.
- Sensation: The “urge” to use the bathroom.
- Balloon Expulsion Test: A balloon is filled with 50cc of water. Healthy controls pass it in <60 seconds. Many PD patients fail this test entirely.
- Defecography (MRI or X-ray): Gel is inserted into the rectum, and the patient evacuates it inside an MRI machine. This provides a total view of all pelvic organs, which is especially useful for patients with concurrent bladder issues.
Stanford Clinical Findings: Gender and Sensation
- Gender Differences in Dysfunction:
- Men: More likely to struggle with muscle relaxation (spasticity) during evacuation.
- Women: More likely to have weaker pelvic floor muscles (low squeeze pressure).
- The Incontinence Paradox: 50% of PD patients with constipation also have “weak squeeze” pressure, putting them at high risk for fecal incontinence.
- Hyposensitivity: One-third (33%) of PD patients suffer from a lack of “urge.” They cannot sense when their rectum is full, leading to significant delays in evacuation.
Researchers hope that the tests will lead to a better understanding of the clinical characteristics of the patients with constipation who have Parkinson’s disease. They hope to learn from the tests and be able to determine the patients who may be developing Parkinson disease in the future based on their differences in the indirect physiology and motility testing.
Questions and Answers for Dr. Leila Neshatian
Q: Did the use of Sinemet (levodopa) affect the study results?
A: About half of the study participants were taking Sinemet. However, the study did not have a large enough sample size (power calculation) to determine if the findings were caused by the medication or the disease itself. Furthermore, it is currently unknown if testing occurred during the “on” or “off” phase of the medication.
Q: Do PD medications cause constipation?
A: Yes. Dopamine agonists are known to delay GI transit, and anticholinergics have a major impact on worsening constipation. However, doctors must balance these side effects against the motor benefits. The goal is to use the “toolbox” of lifestyle changes and GI-specific meds to manage the side effects rather than stopping the PD treatment.
Q: What is the significance of gender differences in PD research?
A: There is a need for more work here. The roles of hormones in both the pathology and the management of PD are not yet fully understood.
Panel with Dr. Kathleen Poston and Dr. Leila Neshatian
Moderated by Dr. Todd Coleman

Q: What do we know about “resilience” in Parkinson’s? Can we predict trajectories for patients who seem to do better than others?
A (Dr. Poston): Resilience is the concept that pathological changes (like alpha-synuclein) have occurred in the brain, but the person remains asymptomatic. The brain essentially finds “redundancies” or new pathways to accomplish tasks.
The Role of Exercise: Exercise is the single most proven factor in building resilience. It helps the brain find new circuits to maintain motor performance despite underlying damage.
GI Resilience: We do not yet have an answer for resilience in the GI tract, but we are investigating whether specific diets, supplements, or microbiome compositions can provide a similar “buffer” against symptoms.
Q: Is there a specific subtype of constipation that points toward a higher risk of developing Parkinson’s?
A (Dr. Neshatian): While 10% of the general population has constipation, 80% of PD patients do. A large case-control study using US insurance claims data suggested that slow transit constipation increases PD risk by approximately 1.5-fold. However, insurance claims data is often unreliable. Our current consortium research aims to use physiological testing to find a more definitive “instinct” or biomarker for future diagnosis.
Q: How do neurological conditions specifically affect gut motility and inflammation?
A (Dr. Neshatian): It is a bidirectional communication involving the vagus nerve and the immune system. Notably, the GI tract produces more serotonin than the brain, which is a key neurotransmitter for both gut and brain function.
Q: There is a lot of controversy regarding the “Braak Hypothesis” and body-first vs. brain-first. What is the latest on how the protein alpha-synuclein travels?
A (Dr. Poston): This remains a “source of uncertainty” among neurologists. This is an important thing for us to understand better
The Pesticide Link: Dr. Bill Langston (now at Stanford) discovered decades ago that certain toxins in pesticides could kill dopamine neurons in mice, creating the first animal model of PD.
The Interplay: While not 100% of cases are “body-first,” there is likely a complex interplay between environmental exposures in the GI tract and genetic predispositions in the brain.
Q: What is the prevalence of SIBO (Small Intestinal Bacterial Overgrowth) in PD, and how is it treated in the context of Parkinson’s?
A (Dr. Neshatian): SIBO is found in over 50% of PD patients (compared to ~30% in the general population) because of abnormal gut motility.
Excessive bacteria in the small intestine can metabolize Levodopa into dopamine before it ever reaches the brain. This causes “delayed-on” or “no-on” medication states and significant fluctuations.
Diagnosis & Treatment: Diagnosis is via a simple breath test (often done at home). While antibiotics are used for treatment, they don’t fix the underlying motility issue, so recurrence is common.
[Dr. Coleman: colleague at Stanford engineered a pill that can be swallowed to capture microbial data from all different parts of the GI tract, including the small intestine.]
Q: How does protein interact with levodopa, and what should patients do?
A (Dr. Neshatian and Dr. Poston): Amino acids from protein and levodopa molecules compete for the same transporters.
Timing: Ideally, take levodopa 1–2 hours before a high-protein meal. Many patients find success by shifting their high-protein intake to dinner time.
Individualization: Some patients are highly sensitive (even an almond can block absorption), while others are not.
Nausea Snack: If you must eat with your meds to avoid nausea, use carbohydrates (like a cracker), as they do not interfere with the transporters.
The Warning: Do not stop eating protein. PD patients need roughly 1g of protein per kg of body weight daily to prevent muscle loss (sarcopenia), which can accelerate the disease and worsen motor complications.
Q: Is there a link between gut function and cognitive impairment?
A (Dr. Poston): This is a major unknown. Most studies look at gut health in relation to motor symptoms, not memory. Our consortium has added virtual cognitive testing (supported by philanthropy) to specifically address whether certain gut signatures predict cognitive decline.
Q: Is the probiotic Lactobacillus Plantarum PS128 “signal” or “noise”?
A (Dr. Neshatian and Dr. Poston): There was a 2021 study with 25 people that showed movement score improvements. That is a “good pilot study,” but it is too small to be definitive. We need larger, placebo-controlled trials.
General Recommendation: Focus on fiber and whole foods first. Fiber acts as a “prebiotic” to restore the microbiome naturally. Stanford is working on an evidence-based review of probiotics to help patients navigate the “noise” of the supplement industry.
Q: Can fecal transplants (FMT) help Parkinson’s?
A (Dr. Poston): We are currently using FMT in mouse models to see how human PD fecal tissue affects them. In humans, we are still a few steps away because we haven’t yet defined what a “positive change” in the gut ecosystem actually looks like.
Q: How does the gut-brain axis differ in non-Lewy body (genetic) forms of PD?
A (Dr. Poston): There’s a very small subset of people with Parkinson’s disease that on autopsy do not have Lewy bodies in their brain. It’s typically associated with certain genetic forms of Parkinson’s disease. Some recent studies have shown that while those individuals don’t have Lewy bodies, there is evidence of some misfolding of alpha synuclein in the brain, so that still could be a misfolded nuclear transmission, but there’s something else that’s keeping that from forming physical Lewy bodies. Still some more work to be done, but it’s important that we think about those individuals within this larger context of the gut brain connection.