
Two leading stem cell researchers shared updates on clinical trials for Parkinson’s Disease (PD) in a January 2026 webinar hosted by the American Parkinson Disease Association (apdaparkinson.org). Dr. Jun Takahashi detailed results from Japan’s induced pluripotent stem cell (iPS cell) transplant trial, while Dr. Claire Henchcliffe discussed her work on stem cell trials using both embryonic stem cells and personalized iPS cell approaches. This webinar and these notes are of interest to anyone curious about where stem cell research stands and what’s involved in these experimental treatments.
Dr. Takahashi, from Kyoto University, explained how iPS cells are created from skin or blood cells, reprogrammed into dopamine-producing neurons, and transplanted into the brain. He shared results from seven patients who received transplants in 2018, with a two-year observation period, including PET scan evidence of new dopamine production and motor improvements in some patients.
Dr. Henchcliffe, from UC Irvine, discussed the exPDite-2 study (expditestudy.com) of embryonic stem cells implanted in 12 patients. Higher-dose patients particularly showed movement improvements. She also briefly mentioned ongoing work with personalized iPS cell approaches and addressed immunosuppression requirements in these trials.
Both researchers emphasized that stem cell treatments target dopamine cell loss, so they work best for patients whose primary symptoms respond well to levodopa. Neither treatment is a cure and they won’t help everyone with PD.
Audience questions covered everything from long-term safety concerns to how the transplanted cells are created, with both doctors providing frank assessments of where the science stands today and what remains unknown.
Watch the full webinar on the APDA YouTube channel:
Dr. Gilbert Hosts: Stem Cells & Parkinson’s Disease
Resources related to the webinar:
- Clinical Trials: Parkinson’s Disease + Stem Cell (NIH)
- Fox Trial Finder (MJFF)
- Understanding Stem Cell Therapy in Parkinson’s Disease Treatment (APDA)
Guest speakers gave presentations followed by a Q&A session. Now, on to my detailed notes. – Kevin
Stem Cells and Parkinson’s Disease
The Panelists

- Rebecca Gilbert, MD, PhD is chief mission officer at the American Parkinson Disease Association and moderated the discussion.

- Jun Takahashi, MD, PhD is a professor at the Center for iPS Cell Research and Application (CiRA) at Kyoto University in Japan. He leads a clinical trial using iPS cell-derived dopamine neurons for PD treatment.

- Claire Henchcliffe, MD, DPhil is a professor of neurology and director of the Norman Fixel Institute for Neurological Diseases at UC Irvine. She has been involved in multiple stem cell clinical trials for PD, including the exPDite-2 study using embryonic stem cells.
Webinar Date: January 15, 2026
Host: American Parkinson Disease Association (APDA)
Summary by: Kevin Boyd, Stanford Parkinson’s Community Outreach
What Are iPS Cells?
Dr. Takahashi explained the basics of induced pluripotent stem cells. iPS cells can be made from skin cells or blood cells. These cells can expand unlimitedly and become a variety of cells like neurons, muscle cells, bones, and blood cells. For PD treatment, researchers use iPS cells to create dopamine-producing neurons.
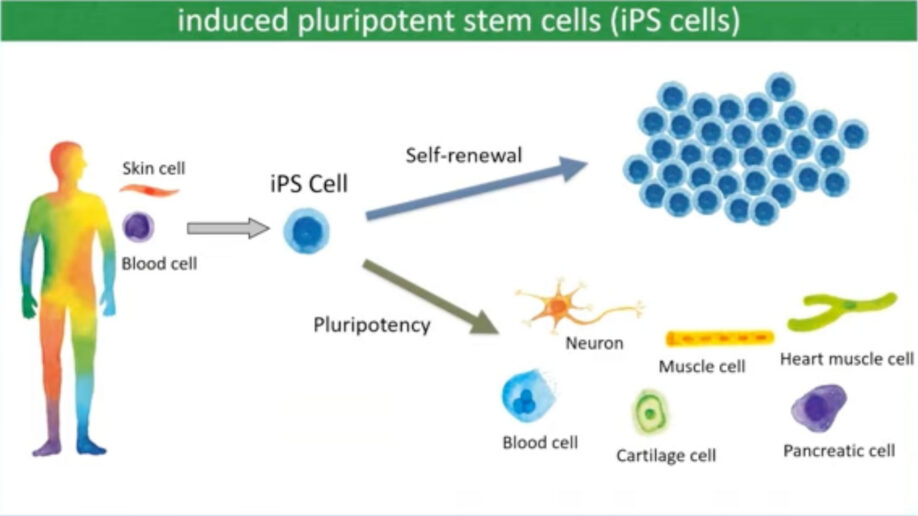
Dr. Takahashi’s team used donor-derived iPS cells for their clinical trial. They obtained blood samples from healthy volunteers at Kyoto University and made clinical-grade iPS cells in their institute, then created dopamine neurons from those iPS cells.
Creating dopamine neurons from stem cells requires careful quality control. Different research teams have developed various production methods – some use purification steps to select only the desired cell types, while others use specific chemicals to prevent cells that might cause tumors from developing. These safety measures help ensure only functional dopamine neurons are transplanted.
iPS cells avoid many ethical concerns associated with embryonic stem cells, which come from embryos.
The Kyoto Trial: Seven Years of Results
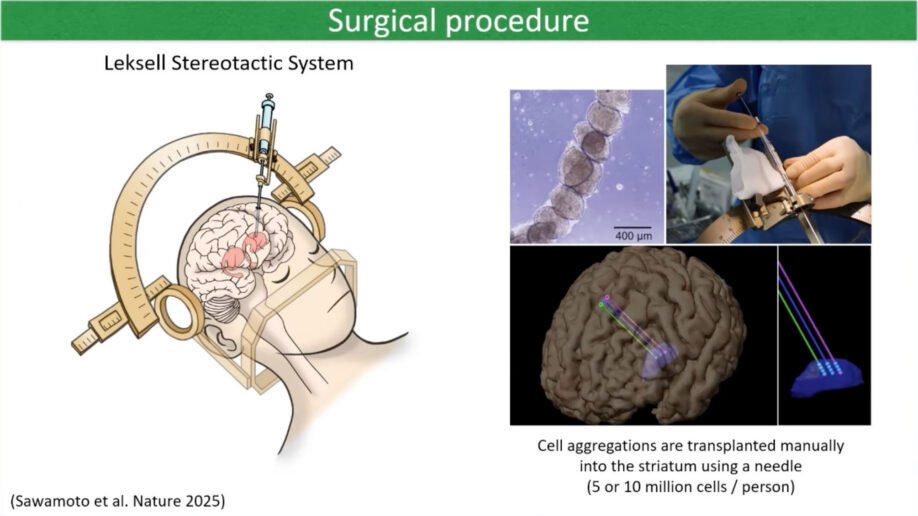
Dr. Takahashi’s team transplanted iPS cell-derived dopamine neurons into seven PD patients in 2018, with an observation period of two years.
The transplant surgery involved placing either five million or ten million dopamine neurons into the putamen, a structure deep within the center of the brain, on each side. PET scan imaging provided objective evidence that the transplanted cells survived and produced dopamine. Dr. Takahashi showed scans demonstrating increased dopamine production in the transplanted regions compared to pre-surgery baseline scans. The dopamine production increased progressively over the first two years after transplant.
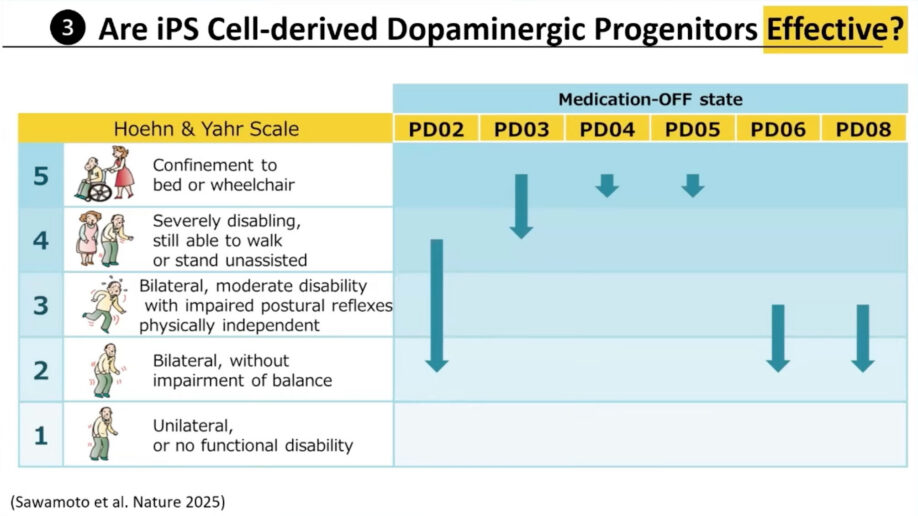
Motor function improvements varied among the seven patients. Dr. Takahashi showed results using the Hoehn and Yahr scale when patients were off medication. Four patients improved one or two stages after two years, but patients four and five did not improve. He noted that patients in later stages might not be good candidates for this type of cell transplantation, though more research with larger samples is needed.
Dr. Takahashi confirmed through this clinical trial that iPS cell-derived dopamine neurons are safe, functional (they survive in the brain and produce dopamine), and effective in improving symptoms for some patients after transplantation. The seven patients encountered no severe adverse events, no graft-induced dyskinesia or disability, and no tumor formation.
Dr. Henchcliffe’s Research

Dr. Henchcliffe began by explaining a 2016 case in Boston involving a single patient who received cells grown from his own skin biopsy. The surgery was performed first at Weill Cornell Medical Center in New York, then six months later at Massachusetts General Hospital. The case was done safely without significant side effects, scans suggested the cells survived, and the patient had some improvement in movement and quality of life.
Dr. Henchcliffe then discussed the exPDite-2 (pronounced “expedite-2”) study, which she’s been working on with a large team. The trial used bemdaneprocel, dopamine precursor cells grown from an embryonic stem cell line that’s been maintained since the late 1990s. Twelve people received transplant surgery with two different cell doses – five people received a lower dose and seven received a higher dose (about three-fold difference). All patients had poorly controlled movement symptoms with medications and had PD for an average of about nine years prior to surgery.
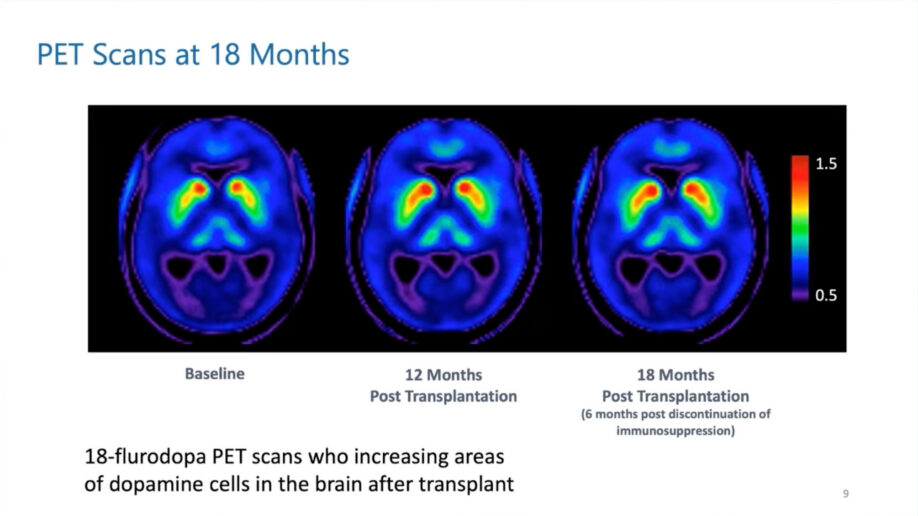
The cells weren’t immunologically matched, so patients received immunosuppression drugs. Follow-up is now over three years with continued safety. PET scans showed encouraging results: at 12 months post-transplant, dopamine cells were filling in, and they continued to fill in at 18 months, even after immunosuppression stopped at 12 months.
Movement measurements at 18 months showed some improvement in patients who received the low dose, with more consistent improvements in the higher-dose group. Dr. Henchcliffe noted they don’t have a placebo group to compare with, so the results could theoretically be placebo effect, but it’s a good first step.
She also mentioned that Aspen Neuroscience has been pursuing a personalized approach, creating iPS cells from each patient’s own tissue. They’ve had seven people undergo transplant and are waiting on results.
Audience Q&A
Q: Can stem cell therapy help patients with atypical Parkinsonism?
A: Dr. Takahashi said the treatment replaces dopamine neurons, so patients need to have Parkinson’s disease, not Parkinson syndromes.
Dr. Henchcliffe added that for conditions like multiple system atrophy (MSA) or progressive supranuclear palsy (PSP), the problem involves more than dopamine cells. People who don’t respond to levodopa probably won’t respond to these cells either. However, she noted there’s hope for stem cell therapies adapted to other Parkinsonisms, mentioning that mesenchymal stem cells have been studied in MSA.
Q: Will the transplanted cells eventually develop Parkinson’s disease themselves?
A: Dr. Takahashi acknowledged this is possible, noting that research on fetal cell transplants showed the grafted cells accumulated alpha-synuclein and Lewy body pathology. So it’s possible the transplanted cells could be affected by Parkinson’s pathology over time.
Dr. Henchcliffe agreed, saying this raises the question of whether combination therapy might eventually be needed, pairing cell transplants with treatments to prevent the spread of alpha-synuclein pathology.
Q: What happens to unused stem cells? Can they cause tumors?
A: Dr. Takahashi said they monitor graft size using MRI and so far there’s no evidence of tumor formation in clinical trials using embryonic stem cells or iPS cell-derived dopamine neurons, though careful observation must continue.
Dr. Henchcliffe added that many transplanted cells die, raising questions about improving cell survival. She noted preclinical work helps researchers feel confident about safety, but monitoring patients closely remains essential. She’s also interested in understanding what happens as immature dopamine cells mature and integrate, and whether researchers can help them survive and mature better.
Q: How can people participate in clinical trials?
A: Dr. Henchcliffe recommended checking clinicaltrials.gov, where all international trials are registered. Users can search by disease (Parkinson’s) and intervention (stem cells) and narrow results by geographic location.
She also mentioned the Michael J. Fox Foundation’s Fox Trial Finder as a matchmaker service for clinical trials. She encouraged people to check their local Parkinson’s centers’ websites and ask their doctors for help finding and contacting trials.
Q: Can patients with deep brain stimulation (DBS) participate?
A: Dr. Takahashi said currently, patients with DBS or who haven’t started levodopa treatment aren’t eligible for these trials.
Dr. Henchcliffe confirmed this, noting several patients with DBS have asked about participating, even offering to have their DBS leads removed, but no one is ready to tackle that yet since it’s early days. She pointed out that some people who received fetal cell transplants decades ago later got DBS, showing the therapies can potentially combine.
Q: Can patients who haven’t started levodopa participate?
Dr. Henchcliffe explained it’s tricky because without an established levodopa response, researchers don’t know if the cells will work. She mentioned that her mentor once suggested stem cell therapy might be especially valuable for young people or those with parkin gene mutations who are at high risk of developing dyskinesia from levodopa, as it could help them avoid levodopa exposure altogether. She noted these are questions for future research about individualizing treatment and determining who truly needs surgery versus who will do fine without it.
Q: In Dr. Takahashi’s trial, could lower-dose patients eventually match higher-dose results?
A: Dr. Takahashi said higher-dose patients produced more dopamine, but even after transplant the dopamine production was still not enough. He explained they started with low doses of cells in this first trial to confirm safety, and now they can increase doses in future trials.
Q: Can stem cell therapy help non-motor symptoms?
A: Dr. Henchcliffe said her trial included non-motor testing to ensure they weren’t harming cognition, but also to look for improvements. She noted that while dopamine delivery targets the motor system, there might be indirect effects similar to those seen with DBS, where improving motor function helps people sleep better and function better. She said nothing jumped out in terms of non-motor improvements in their trial. The treatment is designed to help motor symptoms, but she argued that “if we can help the motor, we free people up to focus on the non-motor.” She thinks future approaches may help symptoms more globally by delivering benefit to broader brain areas in different ways than simply supplying dopamine.
Q: Did anyone in the trials have young-onset PD?
A: Dr. Takahashi said his trial set the eligible age at 50 to 69, so they didn’t include young-onset patients, but after confirming safety and efficacy they could treat young-onset patients in the future.
Dr. Henchcliffe agreed, noting they kept their first trial to those with typical age of onset. She called it a fantastic question because intervening with a one-time treatment for someone facing 50 or 60 years of PD would be exciting, and this population is one researchers are thinking about.
Q: Could focused ultrasound help deliver stem cells?
A: Dr. Takahashi said if you disrupt the blood-brain barrier, he didn’t think it would be good for graft cell survival.
Dr. Henchcliffe said there’s hope for focused ultrasound in delivering biologics like gene therapies and large molecules across the blood-brain barrier, but Dr. Takahashi said that disrupting the barrier exposes cells to vulnerabilities. The blood-brain barrier provides some immune protection, and disrupting it might require treating the transplant more like a solid organ transplant.
Q: What will stem cell therapy cost?
A: Dr. Henchcliffe said that if you could give someone with PD a one-time surgery that works for motor symptoms and avoids the ups and downs of daily life, you have to look at the healthcare costs that are avoided. She noted people could stay in the workforce if they want, remain productive with families, and play with kids and grandkids.
Q: What can researchers learn from each other’s different stem cell approaches?
A: Dr. Takahashi explained that iPS cells and embryonic stem cells have the same characteristics, both expanding limitlessly and differentiating into many cell types. The only difference is that using iPS cells makes autologous transplantation possible, meaning patients can be treated with their own cells without needing immunosuppression. He noted mesenchymal stem cells work differently, providing a cytokine effect like medical treatment rather than replacing lost dopamine neurons.
A: Dr. Henchcliffe said researchers are using cells from different sources and have developed different production methods, with some using purification steps and others using chemicals to avoid cells that might cause tumors. She highlighted the collaboration in the field, noting that teams from Cambridge, Lund, New York, and Irvine have met and helped each other, thinking together about best outcomes, scan types, and surgical approaches.