

In a February 2026 NoSilverBullet4PD webinar, Rick Helmich, MD, with the Donders Institute for Brain, Cognition and Behaviour in The Netherlands, spoke on what he calls “the brain’s backup plan,” of the brain’s compensatory mechanism. This refers to the ability of cortical areas not directly damaged by Parkinson’s Disease (PD) to take over functions that dopamine-dependent circuits can no longer reliably perform. He also identified the two key brain regions involved in this compensation and what causes those regions to fail.
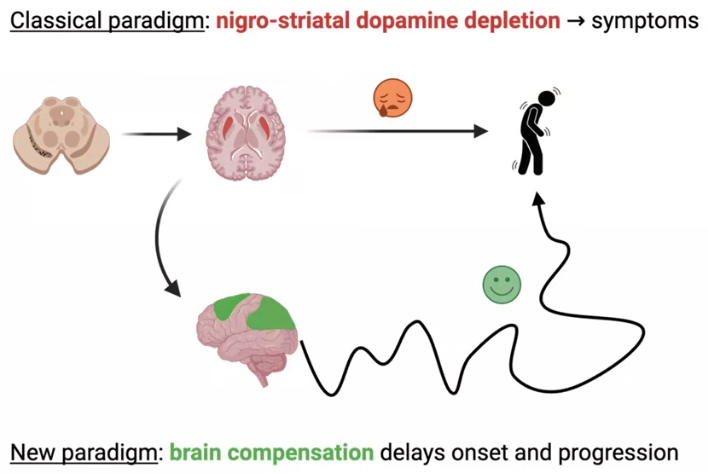
The standard account of PD holds that the death of dopamine-producing cells in the substantia nigra causes the disease’s hallmark symptoms. Helmich said that this explanation, while accurate, leaves important things unexplained. Symptoms don’t appear until more than half of dopamine-producing cells are already gone. The rate of cell loss doesn’t predict how fast someone progresses. And post-mortem studies show dopamine depletion is essentially complete within four years of diagnosis, yet the disease keeps advancing for decades.
As noted, Helmich’s research identifies two brain regions that step up to compensate (the parietal lobe and premotor cortex). While those regions keep working, symptoms worsen more slowly. When those regions begin to fail, symptoms accelerate. Some other takeaways from the talk and Q&A include:
- Exercise and physical activity support mitochondrial function, the energy system that keeps compensatory brain circuits running. This may be one of the most direct links between lifestyle and disease progression.
- Learning new skills stimulates the cortex. Once a skill becomes routine, it migrates to deeper brain structures, so the benefit lies in the novelty and challenge of the learning itself.
- Stress, cognitive overload, and multitasking disrupt the attentional focus that compensatory mechanisms require. Practices like mindfulness may support compensation indirectly by creating the mental conditions it needs.
- The Mediterranean diet has the strongest current evidence for benefit in PD, through two mechanisms: fiber supports gut motility and medication absorption, and reduced inflammation may slow ongoing brain cell damage.
- Levodopa and brain compensation are not in competition. Helmich described them as potentially synergistic, with levodopa supporting deeper dopamine-dependent circuits while cortical compensation engages the outer brain.
Watch the full webinar on NoSilverBullet4PD YouTube channel:
youtube.com/watch?v=UHXxEz88_s4
Now, on to my detailed notes. – Kevin
The Brain’s Backup Plan: The Power of Neuroplasticity in Parkinson’s

Speaker: Rick Helmich, MD, movement disorders neurologist and principal investigator, Donders Institute for Brain, Cognition and Behaviour, Nijmegen, The Netherlands. He leads research into the mechanisms of Parkinson’s, focusing on brain compensation, tremor, and stress.

Moderator: Michel Planquart, NoSilverBullet4PD
Webinar Host: NoSilverBullet4PD, nosilverbullet4pd.com
Webinar Date: February 17, 2026
Summary by: Kevin Boyd, Stanford Parkinson’s Community Outreach
Why Dopamine Loss Doesn’t Tell the Whole Story
The standard account of PD holds that the death of dopaminergic cells in the substantia nigra reduces dopamine in the striatum, a region deep in the brain, producing the hallmark symptoms of slowness, stiffness, and tremor. This reduction is visible on a DaTscan (dopamine transporter scan), which shows progressive loss of dopamine signal in the striatum as the disease advances.
Helmich argued that while this explanation is accurate, it is incomplete. Three lines of evidence point to something the dopamine story alone cannot explain.
First, symptoms do not appear until more than 50% of dopamine cells are already gone (Fearnley and Lees, Brain, 1991). If dopamine loss caused symptoms in a straightforward way, one would expect a more linear relationship between cell loss and clinical presentation.
Second, the rate of dopamine cell loss does not predict how fast symptoms worsen. People whose motor symptoms worsen quickly can show the same degree of dopamine cell loss on DaTscan imaging as people whose motor symptoms worsen more slowly.
Third, post-mortem studies show that dopamine cells are essentially gone within four years of diagnosis, yet symptoms continue to worsen for decades afterward. The disease does not stop advancing when dopamine loss reaches its floor.
Brain compensation, Helmich proposed, is what fills the gap. Brain compensation refers to the ability of brain areas relatively spared by PD to take over functions from damaged circuits.
For compensation to be real and not just coincidental, two things need to be true:
#1 the brain must be responding to something specific (in PD, the loss of dopamine in the deep brain structures that control movement), and
#2 when a compensating area works harder, people must actually function better as a result.
Examples of Compensation
Compensation is not a constant state. Helmich noted that compensatory mechanisms engage when tasks become demanding; routine behavior may not trigger them, but difficulty does. Visual cues and novel physical contexts can serve as that trigger, effectively giving the brain the signal it needs to recruit alternative pathways.
A patient from India with severe freezing of gait could walk normally after his artist daughter painted fake stairs on the floor of his living room, providing the visual information his brain needed to recruit alternative pathways. Other examples Helmich cited included a patient with freezing of gait who could play basketball and another who could walk backward, situations where different brain circuits bridged the gap left by dopamine loss.
Imaging the Backup System
A study from around 2020 found two consistent patterns in those with PD: reduced activity in the basal ganglia, the deep brain structures where dopamine is depleted, and increased activity in regions on the outer surface of the brain not directly affected by dopamine loss. That increased activity in the outer brain is where compensation appears to operate.
To investigate further, Helmich’s team conducted a large fMRI (brain scan) study in which 350 PD patients and 60 healthy controls performed a button-pressing task of varying difficulty while being scanned.
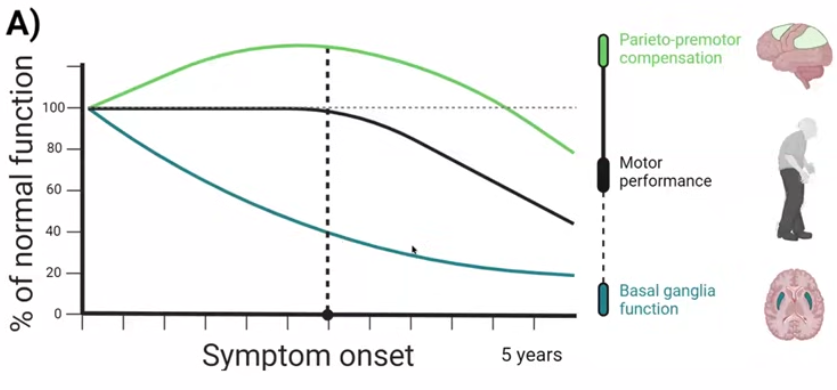
Activity in a brain region called the striatum was reduced in PD patients compared to healthy controls, as expected. But that reduction did not correlate with symptom severity. What did correlate with symptom severity in the PD patients was activity in two cortical regions of the brain: the parietal cortex and the premotor cortex. PD patients with higher activity in these areas had less slowness and stiffness; those with lower activity had more severe symptoms. The same relationship held for cognitive speed. Crucially, PD patients showed more activity in these areas than healthy controls, not merely preserved activity, but genuine hyperactivity. The cortex was working harder to compensate for failing circuits deeper in the brain.
Why the Backup System Eventually Fails
Helmich’s team followed the same PD patient cohort for two years, re-measuring motor symptoms and brain activity. Patients whose symptoms worsened fastest showed the greatest reduction in activity in the parietal and premotor cortex over that period. Patients whose symptoms worsened slowly still had intact activity in those regions after two years. The speed of decline correlated with the degree to which compensation held or failed, not with ongoing dopamine cell loss, which had already reached its floor early in the disease.
Helmich identified three new questions his group is now pursuing:
#1 why does brain compensation fail,
#2 how can it be preserved and protected, and
#3 can it serve as a marker for tracking disease progression?
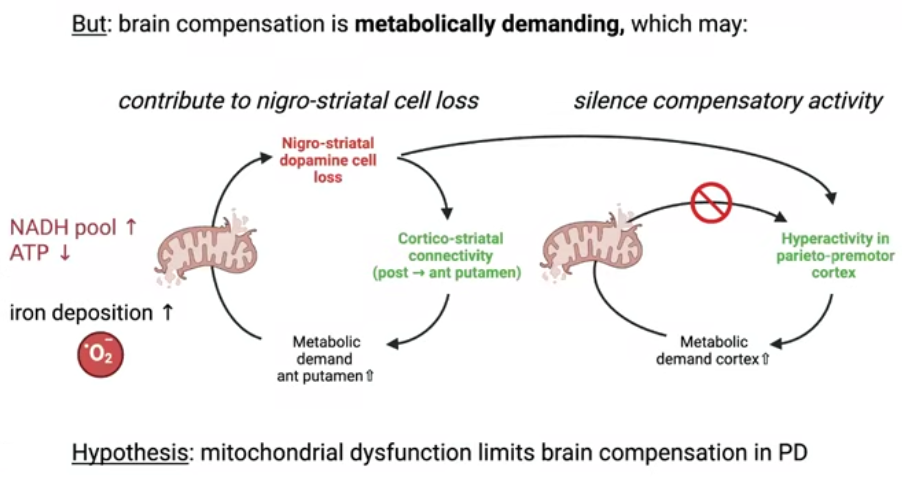
Why does compensation eventually collapse? Helmich offered a hypothesis: brain compensation is metabolically demanding, and the mitochondria, the energy-producing structures in cells, may not be able to sustain the extra work indefinitely. When mitochondrial function falters, compensatory overactivity in the outer brain cannot be maintained.
Approaches to Preserving Compensation
Helmich and colleagues have written about the role of lifestyle interventions in PD, including exercise, stress management, and nutrition, and how they may help protect these brain mechanisms. Exercise and physical activity, in particular, may matter precisely because they support mitochondrial health.
Helmich also mentioned that there could be pharmacological approaches to boosting brain energy but gave no details.
Audience Q&A with Dr. Rick Helmich
When measuring disease progression, were patients on or off medication?
The gold standard for measuring progression is to assess symptoms both after 12 hours off medication and with medication, using a rating scale called the UPDRS (Unified Parkinson’s Disease Rating Scale). Helmich said that symptoms worsened over time under both conditions, and that both measures tracked with the failure of brain compensation. He added that clinical rating scales are a blunt instrument and that his group is working on wearable-based measurement, including smartwatches to continuously track tremor and slowness.
Can EEG be used instead of fMRI to measure cortical activity and track PD progression?
“What you can measure very well with EEG are rhythms, the rhythmic activity of the brain. What you cannot measure with EEG is everything that lies under the cortex. You cannot measure the basal ganglia, you cannot measure the substantia nigra. The big advantage of functional MRI is that you can measure both the deep areas and the more superficial areas at the same time.”
Has microdosing psilocybin or LSD been studied as a way to enhance compensatory mechanisms in PD?
Helmich said there is interest in these substances for their potential effects on mental flexibility, meaning the brain’s ability to adapt and shift its thinking, but that good studies in PD have not yet been done. “It’s an active research field,” he said.
What about neurofeedback as a way to train people to boost their own cortical activity?
Neurofeedback works by measuring brain signals and feeding that information back to the person in real time. “If it works, you can teach people in the scanner to boost their own brain activity,” Helmich said. He cautioned that neurofeedback is technically demanding: movement creates significant signal noise, and learning to influence one’s own brain activity takes many weeks of scanner time. Colleagues at his institute are applying neurofeedback to stress reduction in healthy people, but it has not yet been tried in PD.
Does long-term levodopa use damage brain compensation?
“The short answer is no. I don’t think that levodopa after many years does any harm to the brain. I don’t think levodopa can damage brain compensation. It’s actually the other way around.”
He described levodopa and brain compensation as potentially synergistic: “the combination of dopamine, which is important for the deep areas in the brain, combined with compensatory techniques which involve the superficial layer of the brain, the cortex, that this combination can be synergistic.”
On levodopa’s continued role after four years, when dopamine-producing cells are largely gone: “After four years, your own dopamine production might be gone, but those cells that receive dopamine, so the receptors, are still there. So levodopa continues to play an important role in Parkinson’s disease, but in the later stages, brain compensation plays an additive role.”
Planquart (the moderator) added that levodopa may indirectly support compensation by enabling better exercise performance and by reducing anxiety and stress in some patients.
What nutritional advice would you give?
“The best evidence there is currently is that the Mediterranean diet is beneficial for Parkinson’s. That is probably caused by a few things. One is that it contains a lot of fiber, and fiber is very important for promoting motility of the gut, and that motility is very important for absorbing not only the medication but also other nutrients.”
“The second thing is that a Mediterranean diet indeed reduces inflammation. Inflammation in people with Parkinson’s disease is a little bit increased, not as much as if you have the flu, but just a little bit increased, and we think that contributes to the continuous damage to brain cells.”
What are the implications of this research for deep brain stimulation?
“Deep brain stimulation is particularly suited for small areas because you stick a small electrode into the brain and you can stimulate a few millimeters. It is very effective when a very small area in the brain is malfunctioning, and that is the subthalamic nucleus in Parkinson’s disease. For the cortex, brain stimulation is less suitable because the cortex is much larger. So there we need other techniques, for example, stimulation of the brain via coils on the skull.”
Helmich also confirmed his group is exploring transcranial ultrasound as a non-invasive way to modulate brain activity in cortical targets. He distinguished this from high-intensity focused ultrasound, which concentrates beams at a precise point to raise temperature and create small therapeutic lesions. The lower-intensity approach his group is investigating increases or decreases brain activity without making lesions.
Does individual variation in PD symptoms reflect variation in compensatory capacity?
“I think that the degree to which brain compensation works in Parkinson’s disease differs a lot between people. It might be that in some forms of Parkinson’s disease, the cortex is early on already damaged by the disease itself, so that it cannot compensate. So that could be one reason.”
Do mental exercises like crossword puzzles strengthen compensatory brain areas?
“I think everything that challenges your brain, that keeps you active, helps. If you have a lot of things going on, you have to attend to a lot of things, and your brain is basically overburdened. You cannot focus anymore. Those compensatory mechanisms are wiped away. So you need some focus in order to use these cortical compensatory mechanisms.”
Helmich connected this directly to his stress research: practices like mindfulness that cultivate attentional focus may support compensation by creating the mental conditions it requires.
Are compensatory mechanisms a form of distraction, or focused attention?
“If you look at these areas that are hyperactive in Parkinson’s disease, which are the parietal cortex and the premotor cortex, these are areas that are important for attention, for attentional focusing. Not because they are distracted, but rather the opposite: they can focus their attention on the task they are doing.”
Have you measured amygdala size in PD patients?
Helmich said he has not examined this personally but noted that the amygdala plays a central role in anxiety and stress processing, and that PD patients have elevated rates of both. He said he would expect changes to be present but had no data to confirm.
Can ketogenic diet or intermittent fasting affect neuroplasticity in PD?
Helmich said he is not aware of good studies on this in PD and declined to speculate.
What about red light therapy and photobiomodulation?
“Visual processing plays a role in Parkinson’s. We know that people compensate by using visual cues, for example this staircase that was painted on the floor. So visual processing must play a role in these compensatory mechanisms, same as rhythms and sounds. If red light or certain visual stimulation techniques can actually increase compensation, I don’t know. I don’t think these studies have been done yet.”
Is multitasking good for the brain in PD?
Helmich distinguished between learning new skills and multitasking.
On learning: “I think learning new things is a great way to stimulate your cortex. You first start by activating your cortex to learn it, and when it becomes a routine, those memories are brought down to the deeper parts of your brain.”
On multitasking: “Doing two things at the same time can be challenging because then you might overburden your cortex because it has to switch constantly from one thing to the other. It’s also a source of stress for many of us.”
What is the relationship between the SCAN network and the compensatory parieto-premotor network?
Helmich addressed a recent Nature paper on the SCAN network (somato-cognitive action network), a set of brain regions that links motor and cognitive function, first described in 2023. The paper found that people with PD have abnormally strong connections between their basal ganglia and this network. Helmich said his group examined whether the SCAN network and the compensatory network he described are the same thing and found they are not.
“I think they are two different things: brain compensation and deficits in the SCAN network. But it is indeed highly relevant and interesting to see how they interconnect. I think that will be done in the future.”
Research papers referenced by Dr. Helmich:
- Fearnley JM, Lees AJ. “Ageing and Parkinson’s Disease: Substantia Nigra Regional Selectivity.” Brain, 1991.
- Kordower et al. “Disease Duration and the Integrity of the Nigrostriatal System in Parkinson’s Disease.” Brain, 2013.
- Cabeza et al. “Maintenance, Reserve and Compensation: the Cognitive Neuroscience of Healthy Ageing.” Nature Reviews Neuroscience, 2018.
- Dzialas et al. “Dopamine Transporter Imaging as Objective Monitoring Biomarker in Parkinson’s Disease.” Annals of Neurology, 2025.
- Johansson ME, Toni I, Bloem BR, Helmich RC. “Parkinson’s Disease Progression Is Shaped by Longitudinal Changes in Cerebral Compensation.” Brain, 2026.
- Qu Tian, Erin E. Greig, Christos Davatzikos, Bennett A. Landman, Susan M. Resnick, Luigi Ferrucci. “Higher Skeletal Muscle Mitochondrial Oxidative Capacity Is Associated with Preserved Brain Structure up to over a Decade.” Nature Communications, 2024.
- Trinh J, de Vries NM, Chan P, Dekker MCJ, Helmich RC, Bloem BR. “The Role of Lifestyle Interventions in Symptom Management and Disease Modification in Parkinson’s Disease.” The Lancet, 2025.
Media mentioned by Dr. Helmich: Awakenings (1990 film), based on the work of Oliver Sacks.